
1
METRONIDAZOLE VAGINAL GEL, 0.75%
6111
FOR INTRAVAGINAL USE ONLY
NOT FOR OPHTHALMIC, DERMAL, OR ORAL USE
Rx only
DESCRIPTION
Metronidazole vaginal gel is the intravaginal dosage form of the synthetic antibacterial agent,
metronidazole USP at a concentration of 0.75%. Metronidazole is a member of the imidazole
class of antibacterial agents. Chemically, metronidazole is a 2-methyl-5-nitroimidazole-1-
ethanol.
C
6
H
9
N
3
O
3
M.W. 171.16
Metronidazole vaginal gel is a colorless to yellow gel, containing metronidazole at a
concentration of 7.5 mg/g (0.75%). The gel also contains edetate disodium, hypromellose,
methylparaben, propylene glycol, propylparaben, sodium hydroxide and purified water.
Each applicator full of 5 grams of vaginal gel contains approximately 37.5 mg of metronidazole.
CLINICAL PHARMACOLOGY
Following a single, intravaginal 5 gram dose of metronidazole vaginal gel (equivalent to 37.5 mg
of metronidazole) to 38 healthy female volunteers, a mean maximum serum metronidazole
concentration of 281ng/mL was reported (range: 134 to 464 ng/mL). The average time to
achieve this C
max
was 9.5 hours (range: 4 to 17 hours) after dosing with metronidazole vaginal
gel. This C
max
is approximately 2% of the mean maximum serum concentration reported in
healthy subjects administered a single, oral 500 mg dose of metronidazole (mean C
max
= 12,785
ng/mL).
The extent of exposure [area under the curve (AUC)] of metronidazole, when administered as a
single intravaginal 5 gram dose of metronidazole vaginal gel (equivalent to 37.5 mg of
metronidazole), was 5,989 ng•hr/mL (range: 2,797 to 10,515 ng•hr/mL). This AUC
0-∞
is
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
2
approximately 5% of the reported AUC of metronidazole following a single oral 500 mg dose
of metronidazole approximately 125,000 ng•hr/mL.
Patients with Bacterial Vaginosis
Following single and multiple 5 gram doses of a similar metronidazole vaginal gel product to 4
patients with bacterial vaginosis, a mean maximum serum metronidazole concentration of 214
ng/mL on day one and 294 ng/mL (range: 228 to 349 ng/mL) on day five were reported. Steady
state metronidazole serum concentrations following oral dosages of 400 to 500 mg BID have
been reported to range from 6,000 to 20,000 ng/mL.
Microbiology
The intracellular targets of action of metronidazole on anaerobes are largely unknown. The 5-
nitro group of metronidazole is reduced by metabolically active anaerobes, and studies have
demonstrated that the reduced form of the drug interacts with bacterial DNA. However, it is not
clear whether interaction with DNA alone is an important component in the bactericidal action of
metronidazole on anaerobic organisms.
Culture and sensitivity testing of bacteria are not routinely performed to establish the diagnosis
of bacterial vaginosis. (See INDICATIONS AND USAGE.)
Standard methodology for the susceptibility testing of the potential bacterial vaginosis
pathogens, Gardnerella vaginalis, Mobiluncus spp., and Mycoplasma hominis, has not been
defined. Nonetheless, metronidazole is an antimicrobial agent active in vitro against most strains
of the following organisms that have been reported to be associated with bacterial vaginosis:
Bacteroides spp.
Gardnerella vaginalis
Mobiluncus spp.
Peptostreptococcus spp.
INDICATIONS AND USAGE
Metronidazole vaginal gel, is indicated in the treatment of bacterial vaginosis (formerly referred
to as Haemophilus vaginitis, Gardnerella vaginitis, nonspecific vaginitis, Corynebacterium
vaginitis, or anaerobic vaginosis) in non-pregnant women. There are no adequate and well-
controlled studies of metronidazole vaginal gel in pregnant women.
NOTE: For purposes of this indication, a clinical diagnosis of bacterial vaginosis is usually
defined by the presence of a homogeneous vaginal discharge that (a) has a pH of
greater than 4.5, (b) emits a “fishy” amine odor when mixed with a 10% KOH solution,
and (c) contains clue cells on microscopic examination. Gram’s stain results consistent
with a diagnosis of bacterial vaginosis include (a) markedly reduced or absent
Lactobacillus morphology, (b) predominance of Gardnerella morphotype, and (c)
absent or few white blood cells.
Other pathogens commonly associated with vulvovaginitis, e.g., Trichomonas vaginalis,
Chlamydia trachomatis, N. gonorrohoeae, Candida albicans, and Herpes simplex virus should
be ruled out.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
3
CONTRAINDICATIONS
Metronidazole vaginal gel is contraindicated in patients with a prior history of hypersensitivity to
metronidazole, parabens, other ingredients of the formulation, or other nitroimidazole
derivatives.
WARNINGS
Convulsive Seizures and Peripheral Neuropathy
Convulsive seizures and peripheral neuropathy, the latter characterized mainly by numbness or
paresthesia of an extremity, have been reported in patients treated with oral or intravenous
metronidazole. The appearance of abnormal neurologic signs demands the prompt
discontinuation of metronidazole vaginal gel therapy. Metronidazole vaginal gel should be
administered with caution to patients with central nervous system diseases.
Psychotic Reactions
Psychotic reactions have been reported in alcoholic patients who were using oral metronidazole
and disulfiram concurrently. Metronidazole vaginal gel should not be administered to patients
who have taken disulfiram within the last two weeks.
PRECAUTIONS
Metronidazole vaginal gel results in minimal peak serum levels and systemic exposure (AUCs)
of metronidazole compared to 500 mg oral metronidazole dosing. Although these lower levels of
exposure are less likely to produce the common reactions seen with oral metronidazole, the
possibility of these and other reactions, cannot be excluded presently. Data from well-controlled
trials directly comparing metronidazole administered orally to metronidazole administered
vaginally are not available.
General
Patients with severe hepatic disease metabolize metronidazole slowly. This results in the
accumulation of metronidazole and its metabolites in the plasma. Accordingly, for such patients,
metronidazole vaginal gel should be administered cautiously.
Known or previously unrecognized vaginal candidiasis may present more prominent symptoms
during therapy with metronidazole vaginal gel. Approximately 10% of patients treated with
metronidazole vaginal gel developed symptomatic Candida vaginitis during or immediately after
therapy.
Disulfiram-like reaction to alcohol has been reported with oral metronidazole, thus the
possibility of such a reaction occurring while on metronidazole vaginal gel therapy cannot be
excluded.
Metronidazole vaginal gel contains ingredients that may cause burning and irritation of the eye.
In the event of accidental contact with the eye, rinse the eye with copious amounts of cool tap
water.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
4
Information for the Patient
The patient should be cautioned about drinking alcohol while being treated with metronidazole
vaginal gel. While blood levels are significantly lower with metronidazole vaginal gel than with
usual doses of oral metronidazole, a possible interaction with alcohol cannot be excluded.
The patient should be instructed not to engage in vaginal intercourse, or use other vaginal
products (such as tampons or douches) during treatment with this product.
Metronidazole vaginal gel, 0.75% is supplied with 5 vaginal applicators. For once daily dosing,
one applicator should be used per dose. For twice-daily dosing, the applicator should be washed,
rinsed, and dried following the morning application, and re-used for the evening dose. After the
evening dose, the applicator should be discarded and a new applicator used for the next day’s
dose. See DIRECTIONS FOR USE for complete instructions on how to use the product and the
vaginal applicator.
Drug Interactions
The intravaginal administration of metronidazole vaginal gel, results in relatively lower systemic
metronidazole concentrations compared to that following a 500 mg metronidazole oral dose (see
CLINICAL PHARMACOLOGY). The following drug interactions were reported for oral
metronidazole.
Oral metronidazole has been reported to potentiate the anticoagulant effect of warfarin and other
coumarin anticoagulants, resulting in a prolongation of prothrombin time. This possible drug
interaction should be considered when metronidazole vaginal gel is prescribed for patients on
this type of anticoagulant therapy.
In patients stabilized on relatively high doses of lithium, short-term oral metronidazole therapy
has been associated with elevation of serum lithium levels and, in a few cases, signs of lithium
toxicity.
Use of cimetidine with oral metronidazole may prolong the half-life and decrease plasma
clearance of metronidazole.
Drug/Laboratory Test Interactions
Metronidazole may interfere with certain types of determinations of serum chemistry values,
such as aspartate aminotransferase (AST, SGOT), alanine aminostransferase (ALT, SGPT),
lactate dehydrogenase (LDH), triglycerides, and glucose hexokinase. Values of zero may be
observed. All of the assays in which interference has been reported involve enzymatic coupling
of the assay to oxidation-reduction of nicotinamide-adenine dinucleotides (NAD + NADH).
Interference is due to the similarity in absorbance peaks of NADH (340 nm) and metronidazole
(322 nm) at pH 7.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Metronidazole has shown evidence of carcinogenic activity after chronic oral administration in
mice and rats. Pulmonary tumors and lymphomas were reported in several oral mouse studies in
which mice were dosed at 75 mg/kg and above (about 5 times the clinical human dose based on
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
5
body surface area comparison). Malignant liver tumors were reported in male mice dosed at
doses equivalent to a human dose of 41 mg/kg/day (33 times the recommended clinical dose
based on body surface area comparisons). Chronic oral dosing of metronidazole in rats at doses
above 150 mg/kg (about 120 times the clinical human dose based on body surface area
comparison) has resulted in mammary and hepatic tumors. Two lifetime tumorigenicity studies
in hamsters have been performed and reported to be negative. No life-time studies were
performed to evaluate the carcinogenic potential of metronidazole vaginal gel, 0.75%.
Metronidazole has shown mutagenic activity in a number of in vitro assay systems. In addition, a
dose dependent increase in the frequency of micronuclei was observed in mice after
intraperitoneal injections. An increase in chromosome aberrations has been reported in one study
of patients with Crohn’s disease who were treated with 200 to 1200 mg/day of metronidazole for
1 to 24 months. However, in a second study, no increase in chromosome aberrations was
reported in patients with Crohn’s disease who were treated with metronidazole for 8 months.
Fertility studies have been performed in mice up to six times the recommended human oral dose
(based on mg/m
2
) and have revealed no evidence of impaired fertility.
Pregnancy
Teratogenic Effects
Pregnancy category B
There is no data available regarding the use of metronidazole vaginal gel in pregnant women and
therefore, no adequate and well-controlled studies in pregnant women. Oral reproductive toxicity
studies have been performed in mice at doses up to six times the recommended human dose
based on body surface area comparisons and have revealed no evidence of impaired fertility or
harm to the fetus. However, in a single small study where the drug was administered
intraperitoneally, some intrauterine deaths were observed.
Animal studies have shown that metronidazole crosses the placental barrier and enters the fetal
circulation rapidly. Because animal reproduction studies are not always predictive of human
response, and because metronidazole crosses the placental barrier and is a carcinogen in rodents,
this drug should be used during pregnancy only if clearly needed.
Nursing Mothers
Specific studies of metronidazole levels in human milk following intravaginally administered
metronidazole have not been performed. However, metronidazole is secreted in human milk in
concentrations similar to those found in plasma following oral administration of metronidazole.
Because of the potential for tumorigenicity shown for metronidazole in mouse and rat studies, a
decision should be made whether to discontinue nursing or to discontinue the drug, taking into
account the importance of the drug to the mother.
Pediatric Use
The safety and efficacy of metronidazole vaginal gel in the treatment of bacterial vaginosis in
post-menarchal females have been established on the extrapolation of clinical trial data from
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
6
adult women. The safety and efficacy of metronidazole vaginal gel in pre-menarchal females
have not been established.
Geriatric Use
Clinical studies with metronidazole vaginal gel did not include sufficient numbers of subjects 65
years of age or older to determine whether they respond differently than younger subjects. Other
reported clinical experience has not identified differences in responses between elderly and
younger patients.
ADVERSE EVENTS
Clinical Trials
There were no deaths or serious adverse events related to drug therapy in a clinical trial
involving 220 non-pregnant women who received metronidazole vaginal gel once daily at
bedtime for 5 days. Metronidazole vaginal gel was discontinued in 5 patients (2.3%) due to
adverse events. Adverse events judged definitely or probably related to drug occurred in 25
patients (11.4%).
The incidence of all adverse events was 42% (92/220). Events occurring in ≥ 1% of patients
were: fungal infection (12%), headache (7%), pruritus (6%), abdominal pain (5%), nausea (3%),
dysmenorrhea (3%), pharyngitis (2%), rash (1%), infection (1%), diarrhea (1%), breast pain
(1%), and metrorrhagia (1%).
Adverse events judged to be definitely related or probably related to study medication occurring
in ≥ 1% of patients were: fungal infection (9.1%).
Additional uncommon events, not necessarily related to metronidazole vaginal gel, but reported
by < 1% of those women treated included:
General: allergic reaction, back pain, flu syndrome, mucous membrane
disorder, pain
Cardiovascular: anorexia, constipation, dyspepsia, flatulence, gingivitis, vomiting
Nervous System: depression, dizziness, insomnia
Respiratory System: asthma, rhinitis
Skin and Appendages: acne, sweating, urticaria
Urogenital System: breast enlargement, dysuria, female lactation, labial edema,
leucorrhea, menorrhagia, pyleonephritis, salpingitis, urinary
frequency, urinary tract infection, vaginitis, vulvovaginal disorder
Other Metronidazole Formulations
Other Vaginal Formulations
Other effects that have been reported in association with the use of other formulations of
metronidazole vaginal gel include: unusual taste and decreased appetite.
Topical (dermal) Formulations
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
7
Other effects that have been reported in association with the use of topical (dermal) formulations
of metronidazole include skin irritation, transient skin erythema, and mild skin dryness and
burning. None of these adverse events exceeded an incidence of 2% of patients.
Oral and Parenteral Formulations
Metronidazole vaginal gel results in minimal peak serum levels and systemic exposure (AUC) of
metronidazole compared to 500 mg oral metronidazole dosing. Although these lower levels of
exposure are less likely to produce the common reactions seen with oral metronidazole, the
possibility of these and other reactions cannot be excluded presently. Data from well-controlled
trials directly comparing metronidazole administered orally to metronidazole administered
vaginally are not available.
The following adverse reactions and altered laboratory tests have been reported with the oral or
parenteral use of metronidazole:
Cardiovascular: Flattening of the T-wave may be seen in electrocardiographic tracings.
Central Nervous System: (See WARNINGS.) Headache, dizziness, syncope, ataxia,
confusion, convulsive seizures, peripheral neuropathy, vertigo, incoordination,
irritability, depression, weakness, insomnia.
Gastrointestinal: Abdominal discomfort, nausea, vomiting, diarrhea, an unpleasant
metallic taste, anorexia, epigastric distress, abdominal cramping, constipation, “furry”
tongue, glossitis, stomatitis, pancreatitis, and modification of taste of alcoholic beverages.
Genitourinary: Overgrowth of Candida in the vagina, dyspareunia, decreased libido,
proctitis.
Hematopoietic: Reversible neutropenia, reversible thrombocytopenia.
Hypersensitivity Reactions: Urticaria; erythematous rash; flushing; nasal congestion;
dryness of the mouth, vagina, or vulva; fever; pruritus; fleeting joint pains.
Renal: Dysuria, cystitis, polyuria, incontinence, a sense of pelvic pressure, darkened
urine.
OVERDOSAGE
There is no human experience with overdosage of metronidazole vaginal gel. Vaginally applied
metronidazole gel, 0.75% could be absorbed in sufficient amounts to produce systemic effects.
(See WARNINGS.)
DOSAGE AND ADMINISTRATION
The recommended dose is one applicator full of metronidazole vaginal gel, (approximately 5
grams containing approximately 37.5 mg of metronidazole) intravaginally once or twice a day
for 5 days. For once a day dosing, metronidazole vaginal gel should be administered at bedtime.
HOW SUPPLIED
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
8
Metronidazole vaginal gel, 0.75% is supplied in a 70 gram tube and packaged with 5 vaginal
applicators.
Store at 20
o
to 25
o
C (68
o
to 77
o
F) [See USP Controlled Room Temperature]. Protect from
freezing.
METRONIDAZOLE VAGINAL GEL, 0.75%
DIRECTIONS FOR USE
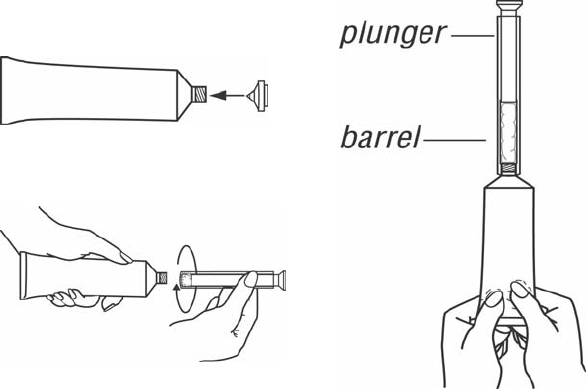
1. Filling the applicator
• Remove cap and
puncture metal seal on
tube with the pointed tip
of cap. (See Figure 1)
• Screw end of applicator
onto tube. (See Figure
2)
• Slowly squeeze gel out
of tube and into
applicator. Plunger will
stop when the applicator
is full. (See Figure 3)
• Unscrew applicator and
replace cap on tube.
Figure 1
Figure 2
Figure 3
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

9
2. Inserting the applicator
• The applicator may be
inserted while lying on
your back with your
knees bent or in any
comfortable position.
• Hold filled applicator
by barrel, and gently
insert into vagina as far
as it will comfortably
go. (See Figure 4)
• Slowly press the
plunger until it stops to
deposit gel into vagina
and then withdraw the
applicator.
Figure 4
3. Care of the applicator
If physician prescribes
once-a-day dosing:
• After use, the
applicator may be
discarded
If physician prescribes
twice-a-day dosing:
• After the morning dose,
pull the plunger out of
the barrel. (See Figure
5)
• Wash both plunger and
barrel in warm soapy
water and rinse
thoroughly and dry.
• For the evening dose,
the applicator can be
reassembled by gently
pushing the plunger
back into barrel.
Figure 5
IMPORTANT:
This product is supplied with 5 vaginal applicators.
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
10
For once-a-day dosing:
• apply one applicator full at bedtime. After using the applicator, discard it and use a
new applicator for the next day’s dose.
For twice-a-day dosing:
• apply one applicator full each morning and evening for five days. Following the morning
application, wash, rinse, and dry the applicator (as instructed above). The applicator may
be reassembled for the evening dose. After the evening dose, discard the applicator and
use a new applicator for the next day’s dose.
WARNINGS:
• If significant irritation develops from the use of this medication, discontinue use and consult
your physician.
• Do not use during pregnancy except under the supervision of a physician.
• Keep this and all medications out of reach of children.
• For vaginal use only. Not for use in the eyes, on the skin or in the mouth.
Store at room temperature. Avoid exposure to extreme heat or cold. See end of carton and
bottom of tube for lot number and expiration date.
Manufactured By:
TEVA PHARMACEUTICALS USA
Sellersville, PA 18960
Iss. 5/2005
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
